Oligonucleotides: A Cornerstone for Therapeutics & More
ISPE
OCTOBER 27, 2022
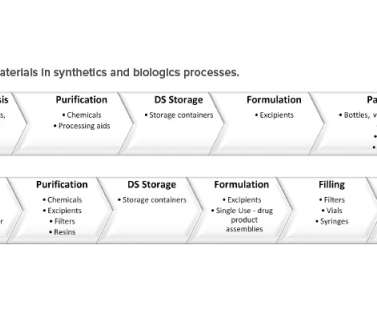
Since 2016, drug approvals per clinical campaign have outpaced the average across all modalities, resulting in 11 FDA-approved therapies in that span. Cone-bottom reaction vessels can provide the turndown required to process a variety of scales and handle different reactions.











Let's personalize your content