FDA Approves Iloperidone for Acute Treatment of Manic, Mixed Episodes in Bipolar I
Drug Topics
APRIL 4, 2024
The drug was previously approved in 2009 to treat schizophrenia.
This site uses cookies to improve your experience. To help us insure we adhere to various privacy regulations, please select your country/region of residence. If you do not select a country, we will assume you are from the United States. Select your Cookie Settings or view our Privacy Policy and Terms of Use.
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.
Used for the proper function of the website
Used for monitoring website traffic and interactions
Cookies and similar technologies are used on this website for proper function of the website, for tracking performance analytics and for marketing purposes. We and some of our third-party providers may use cookie data for various purposes. Please review the cookie settings below and choose your preference.

Drug Topics
APRIL 4, 2024
The drug was previously approved in 2009 to treat schizophrenia.

STAT
MARCH 27, 2025
Both deputy directors at the key Food and Drug Administration center that oversees the regulation of cancer drugs plan on departing the agency, sources told STAT Thursday, highlighting the drain on talent at the FDA created by layoffs, uncertainty, and shifts in policy at the agency even as it is set to lay off thousands more people.
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

STAT
JULY 23, 2024
Shuren, who spent 28 years at the agency, started his FDA career in the Commissioner’s Office in 1998. He became director of the Center for Devices and Radiological Health in 2009 and has served in that position ever since. Continue to STAT+ to read the full story…

STAT
JUNE 28, 2024
Vape manufacturers see the decision as a powerful weapon in their fight against the FDA’s efforts to ban the majority of vapes lining store shelves. The court on Friday overturned a judicial rule, known as the Chevron doctrine, which instructed courts to defer to regulators on certain legal issues.

STAT
SEPTEMBER 26, 2023
Since the Biologics Price Competition and Innovation Act of 2009 created a regulatory pathway for biosimilar medicine review in the U.S., The FDA took great care to apply appropriate scientific rigor to the development and approval standards. Biosimilars are no longer a new and untested class of medicines.

STAT
NOVEMBER 17, 2023
They were approved under the FDA accelerated pathway in 2009 and 2014, respectively, for treating a rare form of blood cancer. The The company’s final study plan was submitted to the FDA in 2022 and is expected to be completed by 2030 , according to FDA briefing documents published earlier this week.

STAT
OCTOBER 16, 2023
The proposal, which is supported by a range of public health groups but fiercely opposed by cigarette makers, is one of the most consequential tobacco policies coming out of the FDA since the agency was given the authority to regulate tobacco in 2009.

STAT
OCTOBER 17, 2024
2 official at the Food and Drug Administration, has spent a lot of time thinking about how to leverage data to improve our health, telling STAT in September that the idea for Highlander Health dates back to 2009. Abernethy, who previously served as the No. Read the rest…

STAT
NOVEMBER 1, 2023
FDA staff said the new type of technology raised concerns about unintended genomic alterations that can potentially cause other side effects, but did not raise any concerns about efficacy. Stelara, introduced in 2009, has been J&J’s top-selling drug since 2019, with sales reaching $9.7 billion in 2022.

PharmaShots
JUNE 1, 2023
Shots: The US FDA has cleared the IND application to initiate a P-II study evaluating PRGN-2009 (off-the-shelf AdenoVerse immunotherapy) + pembrolizumab vs pembrolizumab monotx. while the secondary objectives incl. while the secondary objectives incl.

The FDA Law Blog
FEBRUARY 26, 2024
Karst — While the Biologics Price Competition and Innovation Act (“BPCIA”) is inherently distinct from the Hatch-Waxman Act, many of the fundamental concepts FDA adopted as it enacted the Hatch-Waxman Act made their way into FDA’s implementation of the BPCIA. FDA borrowed this definition from 21 C.F.R.

pharmaphorum
NOVEMBER 28, 2021
Aside from chemotherapy the only improvements to treatment in recent years have included Roche’s VEGF-targeted antibody Avastin (bevacizumab), given a green light by the FDA in 2009, although it was subsequently rejected in Europe on the grounds of a lack of evidence for its efficacy.

The FDA Law Blog
JANUARY 15, 2024
FDA , Petitioners, a liquid nicotine manufacturer, sued FDA arguing that the Agency was arbitrary and capricious in rejecting the Petitioner’s Premarket Tobacco Application (“PMTA”) in violation of the Administrative Procedure Act (“APA”). Youth behavioral data was not specifically required but FDA encouraged such information.

The People's Pharmacy
MAY 8, 2023
Why, then, are FDA inspections abroad so infrequent and disappointing? FDA Inspections Suspended: During the COVID pandemic, the Food and Drug Administration suspended its inspections at most foreign pharmaceutical facilities. Even under the best of conditions, the FDA never inspects all foreign pharmaceutical plants annually.

STAT
APRIL 7, 2023
… Genentech said an internal review of misconduct allegations concerning a landmark 2009 paper co-authored by Marc Tessier-Lavigne, a former top executive at the company and currently president at Stanford University, did not find evidence of fraud or intentional wrongdoing , STAT reports. Well, whatever you do, have a grand time.

The FDA Law Blog
OCTOBER 24, 2023
By Dara Katcher Levy — Yesterday, FDA published a new Draft Guidance, “ Communications from Firms to Health Care Providers Regarding Scientific Information on Unapproved Uses of Approved/Cleared Medical Products Questions and Answers ” (SIUU Guidance or Draft Guidance).

pharmaphorum
APRIL 7, 2021
It was approved by in Europe in late March , after the FDA okayed it for the US market last year. Van de Winkel praised the work of Genentech, the US biotech that was bought by Roche in 2009 for nearly $47 billion, but said he intends to keep Genmab as an independent company.

PharmaShots
JUNE 9, 2023
Shots: The US FDA has accepted an sBLA seeking approval of Keytruda + CT for LA unresectable or metastatic BTC. m-OS (12.7 vs 10.9mos.), 1 & 2 yr. OS rate (52% & 24.9% Your go-to media platform for customized news ranging for multiple indications.

Pharmaceutical Technology
JULY 29, 2022
Janssen’s Risperdal (risperidone), a second-generation antipsychotic , got its first FDA approval for autism-related irritability for children over the age of five, in 2006. Generic versions of both drugs are now available.
Pharmaceutical Technology
OCTOBER 5, 2022
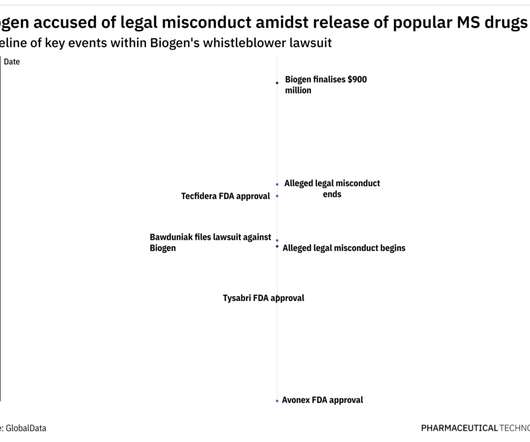
In 2009, former Biogen employee Michael Bawduniak filed a lawsuit claiming that Biogen had violated the False Claims Act and the Anti-Kickback Statute by providing millions of dollars to healthcare providers (HCPs) as an incentive to prescribe three of its multiple sclerosis (MS) drugs. 4 integrins, which play a key role in MS pathology.

The FDA Law Blog
AUGUST 12, 2024
As with other FDA-regulated products, such as human drugs and medical devices, the “regulatory review period” is composed of a “testing phase” and a “review phase.” FDA’s PTE regulations at 21 C.F.R. FDA guidance has been clear that that point for rolling submissions is when the last component has been submitted.

Pharmaceutical Technology
SEPTEMBER 22, 2022
Since 2009, Meiji has marketed tebipenem HBr in Japan. Following receipt of encouraging regulatory feedback from the US Food and Drug Administration (FDA) on the proposed trial design, the company intends to commence the Phase III trial next year. In July, GSK acquired Sierra Oncology for $1.9bn in cash.

The FDA Law Blog
FEBRUARY 2, 2023
Since 1962, the FD&C Act has authorized FDA to require that sponsors of clinical trials submit data from “preclinical tests (including tests on animals)” in order to demonstrate that their drug is safe enough to advance to testing in humans. However, there are signs that FDA is receptive from a policy perspective to alternative methods.

pharmaphorum
DECEMBER 17, 2020
The company’s lead candidate Tenapanor is a targeted, small molecule therapy currently under FDA review. As the co-founder of the biopharma company, Raab has been focusing on advancing patient care for kidney disease since 2009. Patients who undergo dialysis are some of the most fragile.

The FDA Law Blog
OCTOBER 3, 2022
Mr. Bawduniak alleged that, between 2009 and 2014, Biogen paid illegal kickbacks to its largest prescribers to induce them to prescribe the company’s multiple sclerosis drugs, Avonex, Tysabri and Tecfidera, and discourage them from prescribing newer competitor products. This lawsuit was brought to the U.S. See United States ex rel.

The Checkup by Singlecare
SEPTEMBER 13, 2023
In humans, metronidazole is FDA-approved for treating certain bacterial infections. Although not FDA-approved for animals, metronidazole is used off-label in cats when prescribed in veterinary medicine. What is metronidazole used for in cats?

pharmaphorum
DECEMBER 28, 2022
In 2018, over 60% of all new molecular entities came from smaller biopharma firms, compared with just over 30% in 2009. These teams will include experts in regulatory, GMP compliance and quality systems, often including ex-FDA and MHRA inspectors and industry experts.

pharmaphorum
JUNE 28, 2022
The intravenously-administered cephalosporin drug met its objectives in the ERADICATE study in adults with bacterial bloodstream infections caused by Staphylococcus aureus (SAB), setting up an FDA filing before the end of the year, said the Swiss pharma.

The FDA Law Blog
OCTOBER 9, 2022
In 2016, DEA denied two petitions — originally filed in 2011 and 2009 — after HHS concluded that marijuana continued to meet schedule I criteria by having a high potential for abuse, no accepted medical use in the United States, and lacking an acceptable level of safety for use even under medical supervision.

ISPE
SEPTEMBER 15, 2022
First published in 1989, there have been a total of 5 adaptations in 1996, 2003, 2005, 2007 and 2009, but no complete revision. Annex 1 of the EC GMP Guide " Manufacture of Sterile Medicinal Products " has a long history. In 2012, there was a proposal for a complete revision which resulted in a concept paper in 2015.

pharmaphorum
JANUARY 4, 2023
The drug was originally developed by CV Therapeutics – bought by Gilead in 2009 – and first launched in the US in 2008. AmphaStar and Glenmark Pharma also claimed FDA approval for versions of the drug earlier this year. It is sold as Rapiscan in Europe by GE Healthcare.

The People's Pharmacy
MAY 22, 2023
That said, the FDA requires this notification in the official prescribing information: “There have been rare reports of cognitive impairment (e.g., Transient Global Amnesia Can Strike At Any Age: “I suffered from 10 hours of amnesia in 2009 when I was 63 years old. Many people develop cognitive decline regardless of medications.

pharmaphorum
SEPTEMBER 26, 2022
In May, the Delaware court sided with Hospira and ruled that three patents Astellas is relying on for Lexiscan market exclusivity in the US – 8,106,183; 8, 524,883; and RE47301 – are not infringed by Hospira’s generic, which was submitted for FDA approval in 2018.

The FDA Law Blog
FEBRUARY 27, 2024
156, a patent may be extended only once (even if it would be eligible for extension on more than one occasion because it applies to several FDA-approved products), and only one patent may be extended for each regulatory review period. This means that there multiple same-day FDA NDA approvals for the same drug. FDA-2020-E-1840 (Mar.
The Checkup by Singlecare
DECEMBER 5, 2023
Levothyroxine (a thyroid hormone) is FDA approved to treat hypothyroidism , a condition in which the body produces less thyroid hormone than your body needs. It’s FDA approved to treat attention-deficit/hyperactivity disorder (ADHD) symptoms, including inattention, hyperactivity, and impulsivity.

The Checkup by Singlecare
DECEMBER 1, 2023
That is 170% of the daily value (DV) recommended by the US Food and Drug Administration (FDA). Cereal To reduce vitamin D deficiency in America, the FDA now allows more vitamin D to be added to breakfast cereals. The FDA advises adults to have no more than three servings of canned, light tuna per week.

pharmaphorum
MARCH 3, 2021
Symbicort was first FDA-approved in 2006 as a treatment for asthma, followed by an extension to its label covering COPD in 2009. In September last year Mylan and Kindeva argued in the trial that each asserted patent claim is invalid under US patent laws.

PharmaShots
MARCH 29, 2023
Lupin has earned the US FDA approval for the generic version of Banzel as Rufinamide Oral Suspension. Last year TWI announced the US FDA approval of their generic Dexilant is available in 30mg and 60mg capsules which would be released into the market once the patents expire. The exact mechanism of action is uncertain.

The People's Pharmacy
APRIL 3, 2023
Back in 2009, the FDA warned people against using one of the most popular brands, Zicam. The FDA has warned consumers not to use certain Zicam products ( Cold Remedy Nasal Gel , Cold Remedy Nasal Swabs , and kids size Nasal Swabs ). We received a question right around that time. Do you have any suggestions?

Impact Pharmaceutical Services
AUGUST 9, 2022
IMPACT (now part of Syner-G BioPharma Group) has been providing Regulatory Affairs support to a mid-size pharmaceutical company since 2009. The RL was also the primary contact for all correspondence with the FDA related to the program. Regulatory Affairs Strategy and Project Team Lead.

PharmaShots
JUNE 2, 2023
Impact Therapeutics Entered into a License and Collaboration Agreement with Eikon Therapeutics to Develop and Commercialize IMP1734 Date: June 02, 2023 | Tags: Impact Therapeutics, Eikon Therapeutics, IMP1734, Pharma, China, Hong Kong, Macau, Taiwan BMS’ Mavacamten Receives the NICE Recommendation for the Treatment of Obstructive Hypertrophic (..)

pharmaphorum
JANUARY 5, 2021
New product approvals do not seem to have been adversely affected by the pandemic – as at the time of writing, the FDA has approved 48 novel drugs in 2020, matching the 48 medicines defined in the same way in 2019. In 2009, $183m of global market value was due to go off patent in the next five years, in 2019, it was $200m.

pharmaphorum
AUGUST 24, 2022
A federal judge has ruled that a jury trial will be needed to resolve allegations that Indivior attempted to preserve a monopoly by switching to a sublingual film formulation of Suboxone (buprenorphine/naloxone) in 2009 from an older pill version of the product.

pharmaphorum
SEPTEMBER 27, 2022
The suit claimed that Biogen paid millions of dollars to doctors in the form of speaker honoraria, training fees, consulting fees, and meals to induce them to prescribe the MS drugs Avonex (interferon-beta-1a), Tysabri (natalizumab), and Tecfidera (dimethyl fumarate) between 2009 and 2014.
Expert insights. Personalized for you.
We have resent the email to
Are you sure you want to cancel your subscriptions?


Let's personalize your content