FDA-Approved Labeling: Is Enough Enough?
The FDA Law Blog
AUGUST 21, 2023
Livornese — I saw the sign…and the answer is no—FDA-approved labeling apparently is not enough under state failure-to-warn laws, according to certain courts.

The FDA Law Blog
AUGUST 21, 2023
Livornese — I saw the sign…and the answer is no—FDA-approved labeling apparently is not enough under state failure-to-warn laws, according to certain courts.

The FDA Law Blog
FEBRUARY 27, 2023
Palmer — The 2008 Ryan Haight Online Pharmacy Consumer Protection Act placed strict limits on online prescribing or the use of telemedicine encounters to prescribe controlled substances. By Karla L. 829(e); and 2) the modified registration requirement for online pharmacies. 21 U.S.C. § 21 U.S.C. § 802(54)(D)(i).
This site is protected by reCAPTCHA and the Google Privacy Policy and Terms of Service apply.

The Checkup by Singlecare
JANUARY 30, 2024
Hailing from the selective serotonin-reuptake inhibitor ( SSRI ) class of drugs, citalopram is often used for the treatment of major depressive disorder or off-label use treating anxiety disorder. These communication efforts can make all the difference. For all the good work that it does, citalopram also has its downsides.
The Checkup by Singlecare
OCTOBER 30, 2023
In addition, sertraline is prescribed for several other off-label indications, including binge eating disorder and body dysmorphic disorder, even sexual dysfunction conditions like premature ejaculation. Therefore, any onset of symptoms should be managed by immediate communication with a healthcare professional.

ISPE
MARCH 29, 2023
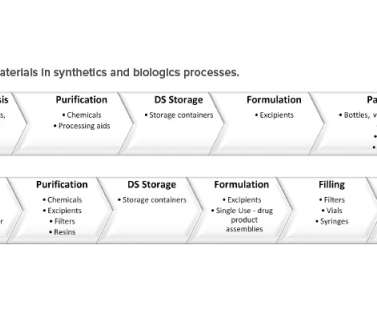
Published June 2008. Challenges with the Current Module 2 Structure The US FDA published a white paper in 2018 calling for a revision to Module 2 because, “there can be a disconnect between applicants and regulators regarding the communication of quality data and its impact on the assessment.

ISPE
MARCH 29, 2023
Published June 2008. Challenges with the Current Module 2 Structure The US FDA published a white paper in 2018 calling for a revision to Module 2 because, “there can be a disconnect between applicants and regulators regarding the communication of quality data and its impact on the assessment.
ISPE
AUGUST 30, 2022
Published 2008. The original molecular weight (MW) specification for polypropylene glycol 2000 (PPG) of 1800–2200 was based on the Food Chemical Codex monograph (90%–110% of label) and not based on a scientific understanding of the process/product requirements. You may unsubscribe from these ISPE communications at any time.
Let's personalize your content